All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Do you know... Which of the following BCMA-directed therapies is NOT associated with CRS and ICANS, due to a lack of direct T-cell activation?
On March 5, 2026, the Multiple Myeloma Hub held a virtual symposium, titled Optimizing dosing of BCMA-directed therapies for improved quality of life in multiple myeloma. During the symposium, Fredrik Schjesvold, Oslo University Hospital, Oslo, NO, provided an overview of B-cell maturation antigen (BCMA)-directed therapies in multiple myeloma (MM).
During this presentation, Schjesvold provides an overview of currently available BCMA-directed treatment modalities, including antibody–drug conjugates (ADCs), bispecific antibodies, and chimeric antigen receptor (CAR) T-cell therapies. Schjesvold outlines differences in mechanisms of action (Figure 1), regulatory approvals, treatment settings, and key efficacy and safety data across approved agents (Figures 2 and 3).
Figure 1. Mechanisms of action of BCMA-directed therapies*
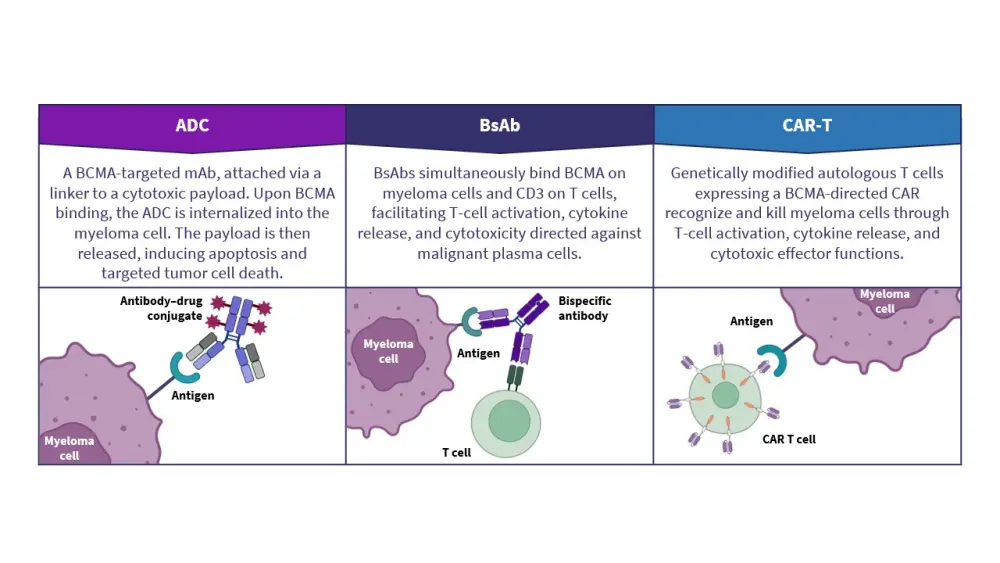
Figure 2. Efficacy of BCMA-directed therapies for multiple myeloma*
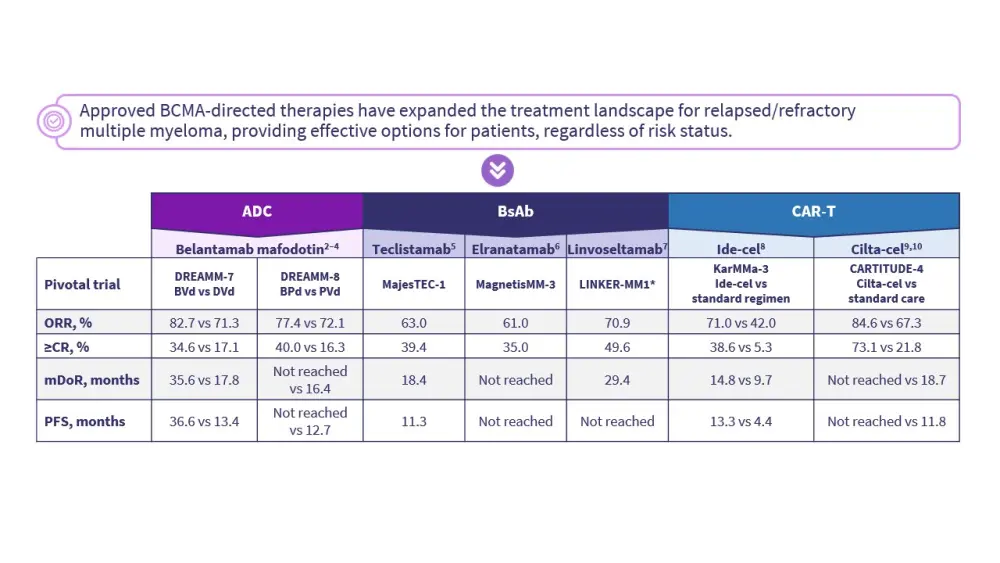
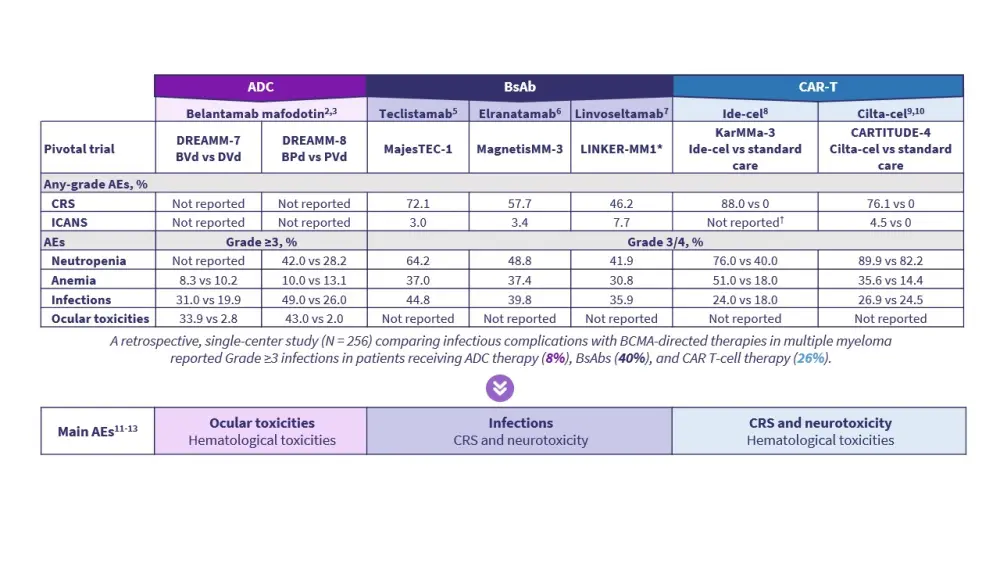
Figure 3. Safety profile of BCMA-directed therapies for multiple myeloma*
Overview of BCMA-directed therapies for multiple myeloma
Overview of BCMA-directed therapies for multiple myeloma
Key points
- BCMA-directed treatment modalities for MM, include ADCs, bispecific antibodies, and CAR T-cell therapies.1
- All BCMA-directed therapies target BCMA on myeloma cells, but they differ in their mechanisms of action for inducing tumor cell death.
- ADCs deliver a cytotoxic payload to BCMA-expressing cells following internalization and do not directly activate T cells.1
- Ocular toxicity is an adverse effect associated with belantamab mafodotin, an ADC, and is generally reversible with dose modification or treatment interruption.2,3
- Bispecific antibodies simultaneously target BCMA on myeloma cells and CD3 on T cells, leading to T-cell activation and tumor cell killing.1
- Cytokine release syndrome (CRS) is commonly observed with bispecific antibodies, particularly early in treatment, and may be mitigated with step-up dosing and supportive care.5-7
- Infections are frequently observed with bispecific antibodies and may require prophylactic strategies, including immunoglobulin replacement and antimicrobial prophylaxis.13
- CAR T-cell therapies involve ex vivo modification of patient-derived T cells to target BCMA, followed by reinfusion as a single treatment.1
- CAR T-cell therapies are associated with CRS and neurotoxicity, with onset typically occurring early following infusion.8-10
- Treatment administration differs across modalities; ADCs and bispecific antibodies require repeated dosing, while CAR T-cell therapies are administered as a single infusion.
- Response rates and progression-free survival vary across treatment modalities and disease settings, with higher response rates generally observed in earlier lines of therapy.
- Infection risk differs between modalities, with prolonged risk observed with bispecific antibodies and a more time-limited risk observed following CAR T-cell therapy.13
- Each BCMA-directed therapy class has a distinct safety profile, including ocular toxicity associated with belantamab mafodotin, and immune-mediated toxicities, which are more commonly observed with bispecific antibodies and CAR T-cell therapies.
- Selection of BCMA-directed therapy requires consideration of efficacy, safety, treatment burden, and patient-specific factors.
This independent educational activity was supported by GSK. All content was developed independently by SES in collaboration with the faculty. The funder was allowed no influence on the content of this activity.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content





