All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
How to transition patients on BCMA-directed bispecific antibody therapy from academic to community care
Do you know... Which of the following adverse events is not associated with B-cell maturation antigen (BCMA)-directed bispecific antibody (BsAb) therapies in multiple myeloma?
Introduction
The relatively recent introduction of B-cell maturation antigen (BCMA)-directed bispecific antibody therapies (BsAbs) into the relapsed/refractory (R/R) multiple myeloma (MM) treatment space has allowed patients to experience therapeutic responses similar to those observed with chimeric antigen receptor (CAR) T-cell therapies but with a simpler “off-the-shelf” administration.1 BCMA is highly expressed on plasma cells, while limited on hematopoietic stem cells and non-hematopoietic tissues, making it a rational target for directed therapies.2 Generally, BsAbs are administered in hospital or academic settings; however, adoption in outpatient and community settings is a growing practice, which subsequently requires standardized guidance for their safe and effective delivery.1,3 Administration of immunotherapies, such as BCMA-directed BsAbs, in a community or outpatient setting has the potential to reduce treatment disparities, lower healthcare costs, and improve patient experience.1 At present, a shared care model that encompasses initiation of therapy in an academic center, followed by transition to community oncology care, is emerging as a feasible and increasingly adopted approach.1,3
Overview of BsAbs for R/R MM
BCMA-directed BsAbs simultaneously bind to BCMA on MM cells and to CD3 on T cells, bringing them into close proximity, and triggering T-cell activation and the release of cytolytic proteins that induce tumor cell apoptosis (Figure 1).2,4
Figure 1. Mechanism of action of BCMA-directed BsAbs*
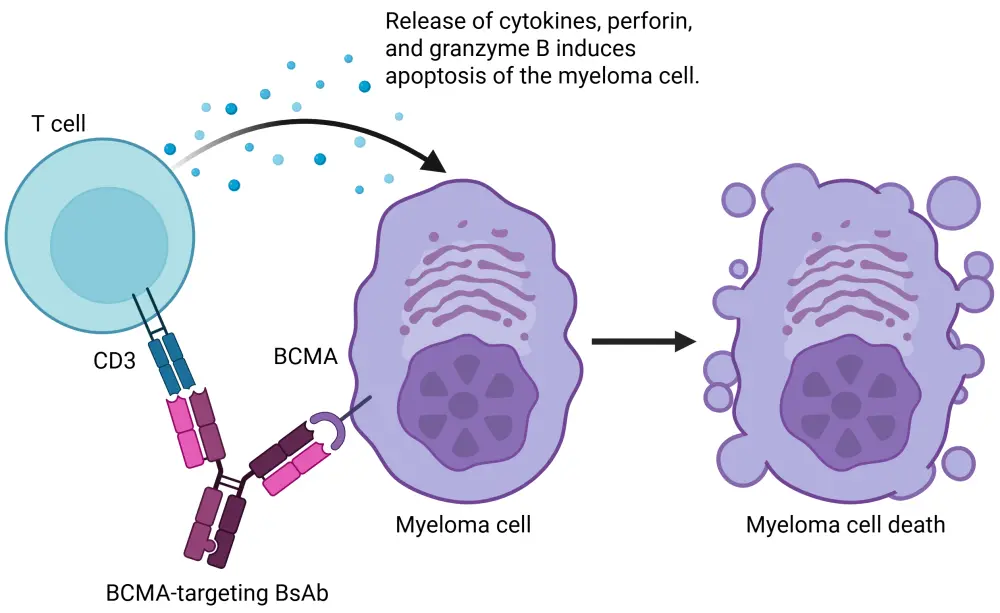
Currently, there are three BCMA-directed BsAbs available for the treatment of R/R MM.5–10 Elranatamab, teclistamab, and linvoseltamab are approved by the European Medicines Agency (EMA) after ≥3 prior lines of therapy and by the U.S. Food and Drug Administration (FDA) after ≥4 prior lines of therapy, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody, for the treatment of R/R MM.5–10 Both elranatamab and teclistamab are subcutaneous therapies, while linvoseltamab is administered intravenously.5–10 Dosing schedules vary with each therapy, although all follow a step-up dosing (SUD) regimen followed by a progressive reduction in dosing frequency.5–10
As with other T cell-targeting agents, key safety considerations with BCMA-directed BsAbs include cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) – both of which are included in boxed warnings in the prescribing information for all three approved therapies – and infection.2
Initiation of therapy
Building a patient care team
Successful administration of BsAbs, particularly in an outpatient setting, should be supported by a multidisciplinary team (MDT) including hematologists/oncologists, nurses, emergency and intensive care specialists, and infectious disease teams.1,11 The MDT should be fully trained on managing BsAb-related toxicities, including CRS and ICANS.1,3 In addition to the MDT, it is recommended that patients receiving BsAbs outside of a care facility have access to a reliable and informed caregiver who can help to monitor AEs and provide additional support throughout SUD and further treatment.1,12
SUD administration in academic centers
SUD during treatment initiation is a prophylactic measure to reduce risk of AEs.13 Patients typically initiate SUD at specialized or academic centers before transitioning to community-based care for ongoing treatment.2,12 Prior to treatment, baseline laboratory data, including complete blood count with differential, a comprehensive metabolic panel, inflammatory markers, and lactate dehydrogenase, should be collected along with the patient’s vital signs.1,13 Given the key safety concerns regarding treatment with BCMA-directed BsAbs, patients receiving teclistamab and elranatamab are typically monitored for approximately 48 hours after early doses, whereas shorter monitoring periods (e.g. ~24 hours) have been used with linvoseltamab.5–10 However, the duration and setting of monitoring may vary.5–10 As the risk of AEs decreases with each BsAb exposure, some centers may consider transitioning patients to community oncology care for the final SUD administration.3
Outpatient SUD administration
In the context of BsAb administration for R/R MM, an outpatient setting can be defined as a healthcare service that does not require an overnight stay in a healthcare delivery institution.3 As the use of BsAbs increases in clinical practice, outpatient SUD administration strategies are being explored to improve patient experience and to reduce healthcare resource utilization.3,13,14 While outpatient administration of BCMA-directed BsAbs during the SUD schedule is feasible, it is not universally practiced and may depend on patient characteristics or infrastructural capabilities.1,3,11,12 Many institutions that use an outpatient SUD scheme prefer to implement a hybrid model in which some step-up doses are administered in an inpatient setting while others are administered in an outpatient setting.11
Monitoring AEs during SUD
Both CRS and ICANS typically occur during SUD or following the first full dose of treatment, so careful monitoring is crucial to ensure AEs are identified throughout this period.3,15 Although SUD itself is used to mitigate risk of treatment-related toxicities, additional strategies may be implemented to further reduce risk, such as prophylactic administration of interleukin-6 receptor antagonists or dexamethasone.2,3,13
If patients are undergoing outpatient SUD, clinicians may consider a period of post-administration observation (e.g. up to 2 hours) to identify early signs of toxicities.13 In some settings, select patients may also be provided with prescriptions for dexamethasone to be taken at home in the event of CRS.13 Following the initial on-site hypersensitivity monitoring, patients may be monitored remotely using technology packages that notify patients to measure vital signs and complete symptom assessments throughout the day; if results are outside of predetermined parameters, the remote devices can alert nurses in the care team to respond.3,12,14
Transition to community oncology care
Unless patients have undergone outpatient SUD, the transfer of patients back to community care may take place following SUD or following 1–3 cycles of treatment depending on whether the patient experienced CRS or neurotoxicity.11,12 To ensure a smooth transition back to community oncology care, effective cross-organizational communication and clear division of responsibilities is paramount.16
Infection prevention
Following the SUD period, ongoing management centers around preventing and monitoring infections.1 Patients with R/R MM are at greater risk of infection if they have undergone multiple lines of treatment, are using immunosuppressive therapies to manage AEs, or have neutropenia or hypogammaglobulinemia.1 An individualized approach should be taken to infection prophylaxis, considering factors such as comorbidities, cytopenias, prior infections, and prior therapy, with the consultation of an infectious disease specialist to guide antimicrobial use.1
Managing AEs
As patients transition from BsAb SUD in an academic setting to BsAb therapy in a community setting, it is crucial that patients and caregivers are educated on the signs and symptoms of treatment-related AEs, including those for CRS and ICANS, and how to appropriately monitor them at home.1–3,13 Being in close contact with the patient care team, and ideally remaining in close proximity to the treatment facility, allows access to appropriate support if needed.1–3,13
Patients typically experience fewer AEs during the maintenance phase of treatment compared with SUD; however, community healthcare teams should be educated on how to manage infection, CRS, and ICANS.3,11 As part of the MDT, community-based nurses can facilitate triage of AE symptoms based on their severity, escalating cases to the physician if needed and advising patients on when to go to the hospital, although action plans may differ between treatment centers (Figure 2).1,12
Figure 2. Triage of AEs following treatment with BCMA-directed BsAbs*
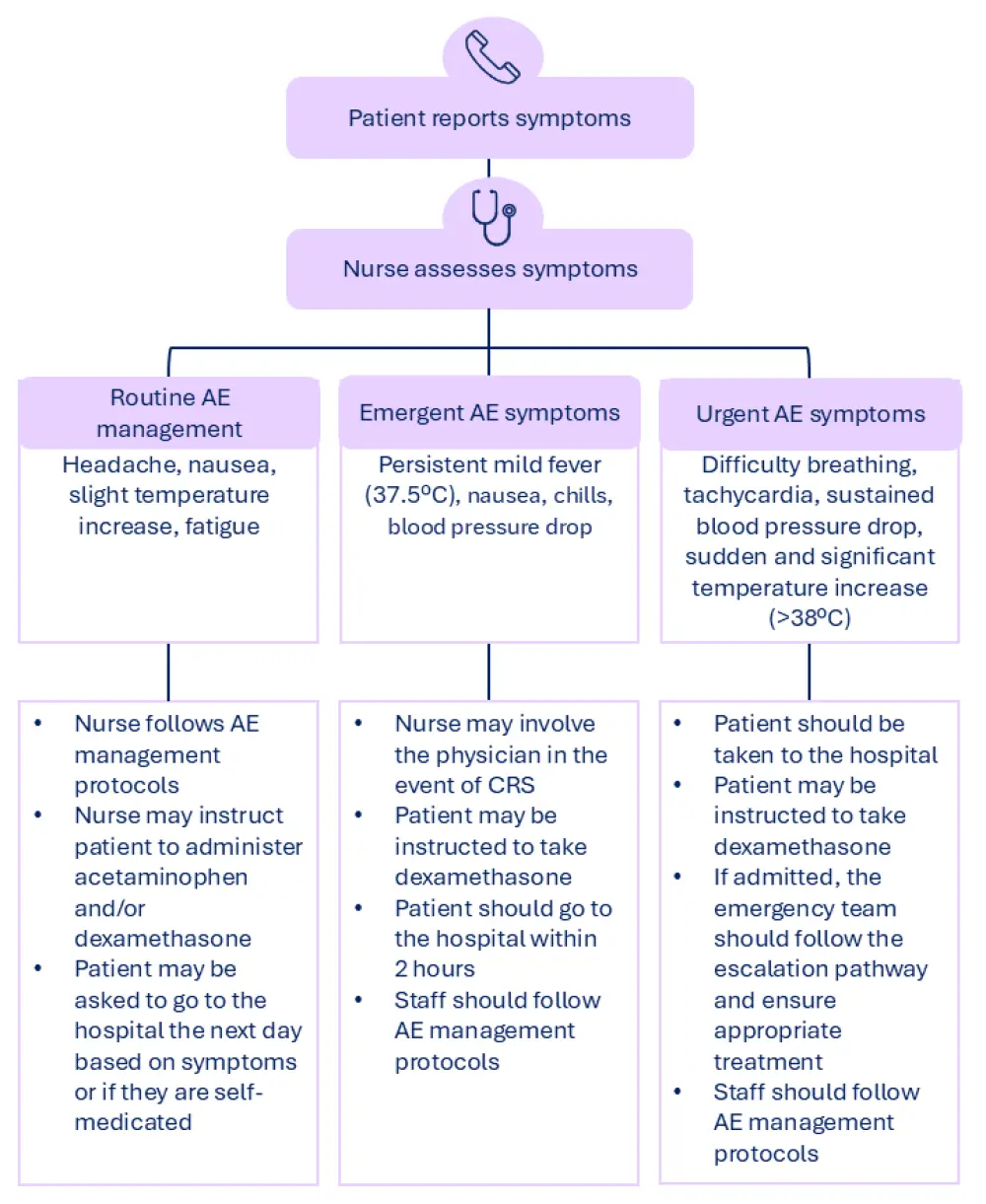
Aside from CRS, ICANS, and infections, BCMA-directed BsAbs may also cause dysgeusia, dry mouth, dysphagia, exfoliation, pruritus, and dry skin.1 Skin-related AEs are generally managed with supportive care measures and may include topical or, in more persistent cases, oral corticosteroids. Oral-related AEs may require additional supportive care and, in select cases, treatment interruption or dose modifications.1 Concerns around hypogammaglobulinemia, lymphopenia, and neutropenia should be considered; unless CRS risk is heightened, growth factors may be used to treat Grade ≥3 neutropenia, while intravenous immunoglobulin replacement can be used to treat hypogammaglobulinemia.12
Operational and infrastructure considerations
Some of the key challenges associated with outpatient administration of BsAbs include transitions from the inpatient to outpatient setting, caring for patients in remote areas, financial burden, managing AEs, and limited experience with the BsAb therapies.3,11,12,17 Additionally, the suitability of an institution to provide, and of a patient to receive, outpatient BsAb therapies can depend on a number of factors (Figure 3).1,3,11,12
Figure 3. Institution and patient criteria for outpatient administration of BsAbs*
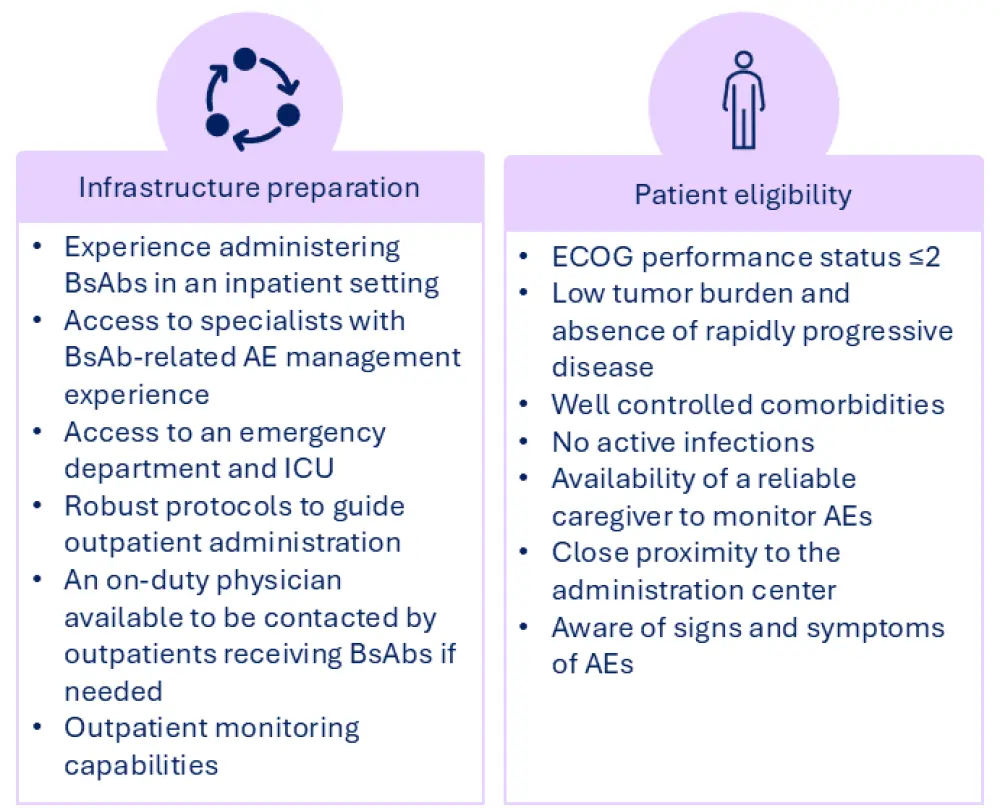
Before considering outpatient administration of BsAbs, cancer care centers should ensure clear workflows are in place for AE management and should implement processes for risk evaluation and mitigation to ensure patient safety during treatment.1,16 Many practices already have the infrastructure in place to implement outpatient administration of other MM treatments, which may be adapted and applied to BsAbs.3 Further to this, existing protocols for CRS management, such as following CAR T-cell therapy, may already be in place and can be repurposed for BsAb treatment.1 Centers should also have a partnership with or have access to an emergency department with adequate training on managing BsAb-related AEs to assist with high-grade toxicities or inpatient needs.1
Future directions
Experience with BsAbs in the outpatient setting varies across care centers, but may expand with the development of appropriate evidence, guidance, and education.3 Reports suggest a recent increase in BsAb referrals to academic centers, indicating increasing comfort with these therapies in the community setting.11 Ongoing dissemination of best practices, guidelines, and training programs between centers may further support the adoption of outpatient administration of BsAbs.3 However, it remains important that physicians and care teams are experienced in treating inpatients with BsAbs first.3 The development of standardized guidelines will also be essential in facilitating an evidence-based approach to outpatient administration of BsAbs and in supplementing local healthcare policies.3,13
In parallel with these implementation considerations, emerging evidence is also informing optimal timing of BsAb use across treatment lines.4,15 It has been suggested that earlier use of BsAbs, in the context of progressive immune dysfunction influenced by prior therapies, may be associated with improved responses and more durable remissions.4,15 In addition, studies have indicated that T-cell fitness may be an effective predictor of responses to BsAbs in earlier treatment settings, although further prospective studies and real-world data are warranted to confirm these findings.15
Conclusions
Currently, BCMA-directed BsAbs represent an effective, “off-the-shelf” therapeutic option for patients with R/R MM.1 While treatment is still commonly initiated in hospital or academic settings due to the risk of AEs, there is an increasing shift toward outpatient and hybrid models with the aim to improve patient experience and reduce healthcare resource utilization, although adoption varies globally due to differences in experience with BsAbs, infrastructure, and the availability of guidelines.3,11–14,17 A crucial consideration when transitioning patients receiving BsAbs from academic centers to community care is the effective monitoring and management of AEs, which requires comprehensive patient/carer education on symptom recognition and clearly defined escalation protocols.1–3,13 As the use of BsAbs in the MM treatment space continues to grow, there is also potential for their implementation in earlier lines of therapy.4,11,15 Continued research, real-world data collection, and sharing of best practices will be key to fully realizing the potential of BsAbs as they become an integral component of the evolving MM treatment landscape.3,13,15
This educational resource is independently supported by Pfizer. All content was developed by SES in collaboration with an expert steering committee. Funders were allowed no influence.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content




