All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
MRD-guided consolidation and treatment de-escalation: Results from the MASTER trial
Minimal/measurable residual disease (MRD) following primary treatment is prognostic of the long-term outcomes in patients with newly diagnosed multiple myeloma (NDMM). However, MRD measurements have not been used to modify therapy. The MASTER trial (NCT03224507) combined daratumumab, carfilzomib, lenalidomide, and dexamethasone (Dara-KRd) to treat patients with NDMM, and used MRD by next-generation sequencing (NGS) to inform the need for and duration of post-transplant therapy.1
Below you will find a summary of the final primary endpoint analysis of the MASTER trial, presented at the 63rd American Society of Hematology (ASH) Annual Meeting and Exposition by Luciano J. Costa.2 Although this study is the first of its kind, there have been significant expectations for it and its practice-changing potential.
Study design
- Patients of all ages were eligible if they had NDMM, a creatinine clearance of ≥40 ml/min, adequate liver and heart function, and an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2.
- There was a planned enrichment of patients with high-risk cytogenetic abnormalities (HRCAs). HRCAs are defined as a gain or amplification of 1q, t(4;14), t(14;16), t(14;20), or del(17p).
Treatment
Figure 1. Treatment schedule for the MASTER trial*
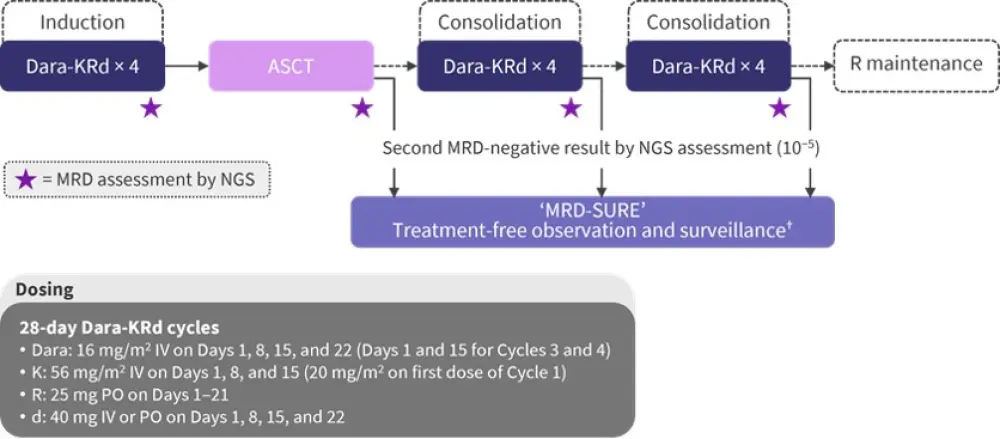
ASCT, autologous stem cell transplantation; d, dexamethasone; Dara, daratumumab; IV, intravenous; K, carfilzomib; MRD, minimal/measurable residual disease; NGS, next-generation sequencing; PO, oral; R, lenalidomide.
*Adapted from Costa2 and data obtained from Costa et al.1
†Patients were evaluated every 8 weeks for the first 24 weeks and every 16 weeks thereafter, and MRD assessment was performed 24 and 72 weeks after completing therapy.
Endpoints
- The primary endpoint was MRD negativity (<10−5 according to the International Myeloma Working Group [IMWG] criteria) as assessed by NGS in patients treated with Dara-KRd, ASCT, and response-adapted Dara-KRd consolidation.
- Secondary endpoints included complete response, and the exploratory endpoint was MRD <10−6.
Results
The study enrolled 123 patients from five sites between March 2018 and September 2020. Patient characteristics are presented in Table 1. The median follow-up was 23.8 months, and MRD status was reported in 118 patients (95%). Primary endpoint results are presented in Figure 2.
Table 1. Patient characteristics*
|
ECOG, Eastern Cooperative Oncology Group; HRCA, high-risk cytogenetic abnormality; LDH, lactate dehydrogenase; R-ISS, revised International Staging System; ULN, upper limit of normal. |
||||
|
Characteristics, % (unless otherwise stated) |
Standard risk |
High-risk |
Ultra high-risk |
Total |
|---|---|---|---|---|
|
Female |
38 |
48 |
46 |
43 |
|
Age |
|
|
|
|
|
Range, years |
36–79 |
35–77 |
41–72 |
35–79 |
|
Age ≥70 years |
23 |
22 |
8 |
20 |
|
Race/ethnicity |
|
|
|
|
|
White |
79 |
72 |
79 |
76 |
|
Racial/ethnic minorities |
21 |
28 |
21 |
23 |
|
ECOG 0–1 |
79 |
87 |
71 |
80 |
|
LDH ≥ULN |
15 |
26 |
25 |
21 |
|
Cytogenetic abnormality |
|
|
|
|
|
Hyperploidy |
51 |
44 |
17 |
41 |
|
del(13q) |
36 |
44 |
75 |
46 |
|
Gain/amplification of 1q |
0 |
52 |
83 |
36 |
|
del(1p) |
6 |
9 |
21 |
10 |
|
t(11;14) |
14 |
15 |
0 |
17 |
|
t(4;14) |
0 |
17 |
54 |
17 |
|
t(14;16) |
0 |
4 |
17 |
5 |
|
del(17p) |
0 |
26 |
58 |
21 |
|
R-ISS III |
2 |
26 |
46 |
20 |
Figure 2. Primary endpoint: MRD negativity (<10−5) at different timepoints
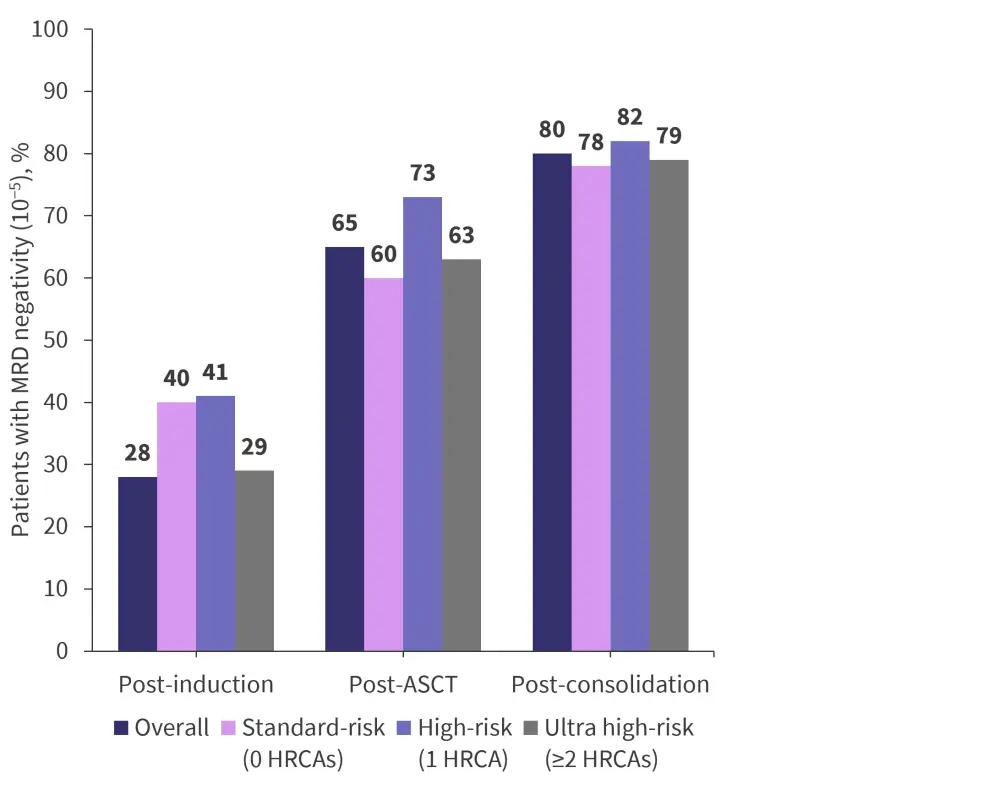
ASCT, autologous stem cell transplantation; HRCA, high-risk cytogenetic abnormality; MRD, minimal/measurable residual disease.
Exploratory analyses
MRD <10−6 negativity
- Overall, 66% of patients reached MRD negativity (<10−6) after MRD-guided consolidation. Rates of MRD negativity at <10−6 were lower than those at a sensitivity of <10−5 and took longer to achieve.
-
- In total 64%, 73%, and 58% of patients with 0, 1, and ≥2 HRCAs reached MRD negativity (10−6) after consolidation, respectively.
- There was no particular subset of patients that was more likely to reach either of the MRD thresholds.
Best IMWG response by phase of therapy in the intention-to-treat group
- A total of 88% of patients achieved a very good partial response (VGPR) after induction (Cycle 4).
- VGPR or better was reached by 98% of patients at the end of consolidation.
Survival endpoints
- The 2-year progression-free survival rate was 91% in patients with no HRCA, 97% in patients with one HRCA, and 58% in patients with ≥2 HRCAs.
- The 2-year overall survival rate was 96% in patients with no HRCA, 100% in patients with one HRCA, and 76% in patients with ≥2 HRCAs.
MRD-SURE subset
- The median follow-up for the MRD-SURE subset was 14.2 months.
- MRD-SURE status was reached by 71% of patients overall; 62% of patients with no HRCA, 78% of patients with one HRCA, and 63% of patients with ≥2 HRCA.
- The risk of MRD resurgence or progression 12 months after treatment discontinuation was 4% in patients with no HRCA, 0% in patients with one HRCA, and 27% in patients with ≥2 HRCAs.
- There were no reported deaths due to MM progression for patients in the MRD-SURE subset.
Safety
Treatment-related severe adverse events were reported in 22 patients (18%), the most common being pneumonia (n = 8) and pulmonary embolism (n = 3).1 Three patients died during treatment. All adverse events are shown in Table 2.
Table 2. Adverse events reported in >25% of patients*
|
*Adapted from Costa.2 |
||
|
Adverse events, % |
Any grade |
Grade ≥3 |
|---|---|---|
|
Any |
100 |
74 |
|
Hematologic |
|
|
|
Neutropenia |
41 |
35 |
|
Lymphopenia |
28 |
22 |
|
Non-hematologic |
|
|
|
Fatigue |
55 |
9 |
|
Bone pain |
55 |
6 |
|
Rash maculo-papular |
41 |
4 |
|
Nausea |
40 |
0 |
|
Constipation |
39 |
0 |
|
Upper respiratory infection |
37 |
1 |
|
Diarrhea |
35 |
4 |
|
Insomnia |
28 |
2 |
|
Infusion-related reaction |
28 |
2 |
|
Dyspnea |
28 |
2 |
|
Cough |
26 |
0 |
|
Hypertension |
26 |
10 |
Conclusion of the MASTER trial
In summary, induction therapy with Dara-KRD, followed by ASCT, MRD-guided consolidation, and treatment cessation was feasible in ~96% of patients, with 72% reaching an MRD-free, treatment-free state (‘MRD-SURE’). Patients with standard and high-risk NDMM had comparable depths of response and a low risk of MRD reappearance or progression when treated with Dara-KRd and ASCT, and therapy was stopped according to MRD status. The quadruplet treatment combined with MRD assessment may provide an alternative treatment strategy, which offers the prospect of a sustained deep response without indefinite maintenance therapy. However, there is still an unmet need for a consolidation treatment option that can clear MRD and improve outcomes in patients with ultra-high risk MM.
Future perspectives
Costa also presented the design of the MASTER 2 trial. The design of MASTER 2 will include the quadruplet therapy as induction, but extending it to six cycles, and using MRD as a decisive factor for further treatment. MRD-negative patients will be randomized between an additional three cycles of quadruplet therapy versus ASCT, followed by 1-year maintenance therapy with an anti-CD38 monoclonal antibody plus lenalidomide. Conversely, patients who remain MRD-positive after induction will be given ASCT consolidation and offered early deployment of a T-cell redirecting therapy versus anti-CD38 combined with lenalidomide as consolidation and maintenance. For all patients, the need of further maintenance therapy beyond 1 year will be decided upon using MRD status, as in the initial MASTER trial.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

