All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Selinexor combined with bortezomib + dexamethasone: the BOSTON trial
The proteasome inhibitor, bortezomib, has demonstrated clinical efficacy in combination regimens for MM. However, prolonged treatment with bortezomib is associated with high rates of peripheral neuropathy (PN). The phase III BOSTON trial (NCT03110562) aimed to compare the efficacy of selinexor + bortezomib + dexamethasone (SVd) with bortezomib + dexamethasone (Vd) and to determine if SVd reduces the rate of PN compared with Vd alone in patients with relapsed/refractory multiple myeloma (RRMM).
At this year’s virtual ASCO Annual Meeting, Meletios A. Dimopoulos outlined the initial results, summarized below.1
Study design
- Primary endpoint: progression-free survival (PFS)
- Secondary endpoints: overall response rate (ORR), ≥ very good partial response (VGPR), Grade ≥ 2 PN
- Adult patients with progressive measurable MM (N = 402) with 1–3 lines of prior therapy underwent 1:1 randomization to receive
- SVd (n = 195) or
- Vd (n = 207)
- Treatment regimens are presented in Table 1
Table 1. Dosing schedules of SVD vs Vd1
|
SVd, selinexor + bortezomib + dexamethasone; Vd, bortezomib + dexamethasone *35-day cycles †Vd biweekly 21-day cycles (Cycles 1–8); Vd weekly 35-day cycles (Cycles ≥ 9) |
||
|
|
Regimen |
|
|
SVd* |
Vd† |
|
|
Selinexor, 100 mg orally
|
Days 1, 8, 15, 22, 29 |
— |
|
Dexamethasone, 20 mg orally |
Days 1, 2, 8, 9, 15, 16, 22, 23, 29, 30 |
Cycles 1–8: Days 1, 2, 4, 5, 8, 9, 11, 12 Cycles ≥ 9: Days 1, 2, 8, 9, 15, 16, 22, 23, 29, 30 |
|
Bortezomib, 1.3 mg/m2 SC
|
Days 1, 8, 15, 22 |
Cycles 1–8: Days 1, 4, 8, 11 Cycles ≥ 9: Days 1, 8, 15, 22 |
Results
- Patient characteristics are presented in Table 2
Table 2. Baseline characteristics of patients enrolled in the BOSTON study1
|
Characteristic |
SVd (n = 195) |
Vd (n = 207) |
|
PR, partial response; SVd, selinexor + bortezomib + dexamethasone; Vd, bortezomib + dexamethasone * del (17p), t(14;16), t(14;14) or amp 1q21 † Patients previously exposed to proteasome inhibitors should have achieved at least a PR |
||
|
Median age, years (range) |
66 (40–87) |
67 (38–90) |
|
Male, % |
59 |
56 |
|
Median time from diagnosis, years (range) |
3.8 (0.4–23) |
3.6 (0.4–22) |
|
High-risk cytogenetics*, % |
50 |
46 |
|
Creatinine clearance 30–60 mL/min, % |
27 |
29 |
|
Number of prior lines of therapy, % 1 2 3 |
51 33 16 |
48 31 21 |
|
Prior treatment, % |
|
|
|
Bortezomib† |
68.7 |
70.0 |
|
Carfilzomib† |
10.3 |
10.1 |
|
Lenalidomide |
39.5 |
37.2 |
|
Daratumumab |
5.6 |
2.9 |
Efficacy
- When compared to Vd, SVd was associated with significantly improved
- response rates in the overall population and by patient subgroup and PFS (Table 3)
- depth of response (Figure 1; ≥ VGPR, p = 0.0082)
- When patients in the control arm (Vd) presented a confirmed progressive disease, they were offered to cross over to a selinexor-based treatment
- Although SVd was associated with lower overall PN rates vs Vd (p = 0.001), PN remained the most common AE resulting in treatment discontinuation:
- SVd: 4.6%
- Vd: 7.4%
Table 3. Patient outcomes to SVd vs Vd1
|
HR, hazard ratio; PFS, progression-free survival; SVd, selinexor + bortezomib + dexamethasone; Vd, bortezomib + dexamethasone * Data cutoff: February 18, 2020. |
|||
|
Patient outcome* |
SVd (n = 195) |
Vd (n = 207) |
p value |
|
Median follow-up, months |
13.2 |
16.5 |
— |
|
PFS, months |
13.93 |
9.46 |
HR 0.70, 0.0066 |
|
ORR, % |
76.4 |
62.3 |
0.0012 |
|
Aged ≥ 65 years |
76.1 |
64.4 |
0.0243 |
|
High-risk cytogenetics |
77.3 |
55.8 |
0.0008 |
|
Creatinine clearance 30 – 60 mL/min |
79.2 |
56.7 |
0.0055 |
|
1 prior line of therapy |
80.8 |
65.7 |
0.0082 |
|
Prior bortezomib treatment |
77.6 |
59.3 |
0.0005 |
|
Prior lenalidomide treatment |
67.5 |
53.2 |
0.0354 |
|
Median duration of response, months |
20.3 |
12.9 |
— |
Figure 1. ORRs to SVd vs Vd in the overall population1
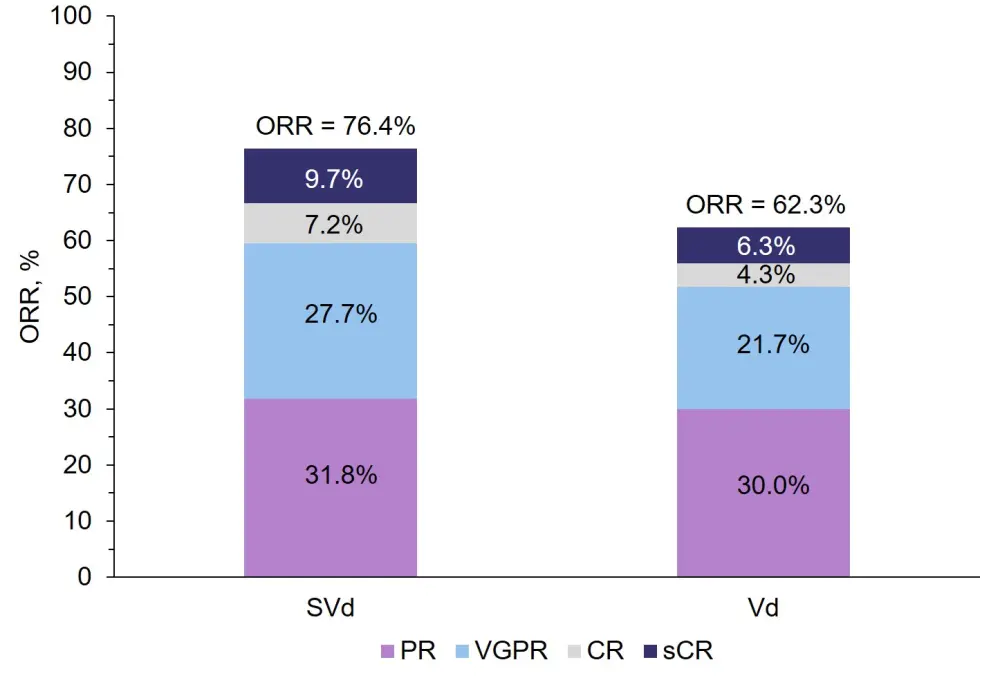
CR, complete response; ORR, overall response rate; PR, partial response; sCR, stringent complete response; SVd, selinexor + bortezomib + dexamethasone; Vd, bortezomib + dexamethasone; VGPR, very good partial response
Safety
- Treatment-related adverse events (TRAEs) were manageable and reversible
- To date, 17% and 11% of patients discontinued SVd and Vd, respectively, due to AEs or toxicities
- Common Grade 3–4 TRAEs are outlined in Table 4
Table 4. Common Grade 3–4 TRAEs observed in > 5% of patients receiving SVd vs Vd1
|
TRAE, treatment-related adverse event; SVd, selinexor + bortezomib + dexamethasone; Vd, bortezomib + dexamethasone |
||
|
Grade 3–4 TRAEs |
SVd (n = 195) |
Vd (n = 207) |
|
Thrombocytopenia |
39.5 |
17.2 |
|
Anemia |
15.9 |
9.8 |
|
Fatigue |
13.3 |
1.0 |
|
Neutropenia |
8.7 |
3.4 |
|
Cataract |
8.7 |
1.5 |
|
Asthenia |
8.2 |
4.4 |
|
Nausea |
7.7 |
0 |
|
Diarrhea |
6.2 |
0.5 |
Conclusions
QW SVd demonstrated significantly superior efficacy over BIW Vd, reducing the risk of progression or death by 30%, and standing as a novel, IMiD-free triplet therapy for patients with RRMM. QW administration of SVd has the potential to reduce hospital visits by up to 40% while also reducing levels of bortezomib-associated PN.1,2
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

