All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Selinexor combination therapy granted full marketing authorization in the UK
On February 21, 2023, selinexor in combination with bortezomib and dexamethasone (XVd) was granted full marketing authorization from the UK Medicines and Healthcare Products Regulatory Agency (MHRA) for the treatment of patients with multiple myeloma (MM) who have received at least one prior line of therapy.1 European marketing authorization was granted in July 2022 based on data from the randomized phase III BOSTON trial (NCT03110562), results of which were previously reported on the Multiple Myeloma Hub.
Here, we summarize additional analyses from this pivotal trial that may further elucidate which patients could benefit most from this regimen. The investigators highlighted the convenience of this combination, which involves a reduced total dose of bortezomib and dexamethasone and fewer hospital visits compared with the standard bi-weekly bortezomib plus dexamethasone (Vd).2
BOSTON trial: subgroup analysis
Prior lines of therapy2
The BOSTON trial included a prespecified analysis to investigate the influence of prior lines of therapy on the efficacy of XVd combination treatment compared with Vd. Patient subgroups included prior lenalidomide therapy, proteasome inhibitor (PI) therapy, immunomodulatory therapy, and prior autologous hemopoietic stem cell transplantation.
This subanalysis showed that XVd therapy was associated with higher overall response rates across all subgroups. However, patients with only one prior line of therapy, those who were lenalidomide naïve, and those previously treated with a PI had significantly higher rates of very good partial response or better. Of note, patients previously exposed to a PI had to have achieved at least a partial response, a 6-month treatment-free interval, and must not have discontinued treatment due to an adverse event.
Median progression-free survival was significantly longer in all subgroups, with the exception of those previously treated with a PI. Overall survival (OS) was favorable across the entire patient cohort when treated with selinexor combination therapy (Hazard ratio [HR], 0.84; 95% confidence interval [CI], 0.57–1.23; p = 0.19).
Grade ≥3 adverse events and discontinuations due to adverse events were more frequent with XVd; however, they were deemed to be generally well-managed. In contrast, Grade 2 peripheral neuropathy was less frequent with weekly XVd versus bi-weekly Vd across all subgroups.
Cytogenetic risk3
Among patients enrolled in the BOSTON trial, 141 had high-risk cytogenetics and 261 were classified as standard-risk cytogenetics. Fluorescent in situ hybridization was performed at screening on collected bone marrow aspirates. Patients were defined as high risk if they had amplification (≥4 copies) of 1q21 in at least 10% of screened plasma cells or one or more of the following cytogenetic abnormalities:
- del(17p)
- t(4;14)
- t(14;16)
Standard-risk patients were those with all other known or unknown baseline cytogenetics. Subanalysis was performed to assess the efficacy of XVd compared with Vd in high- and standard-risk patients.
Median progression-free survival and time to next treatment were longer in both the standard- and high-risk groups when treated with selinexor-based triplet compared with Vd (Figure 1).
Figure 1. Median progression-free survival and time to next treatment in A high-risk patients B standard-risk patients*
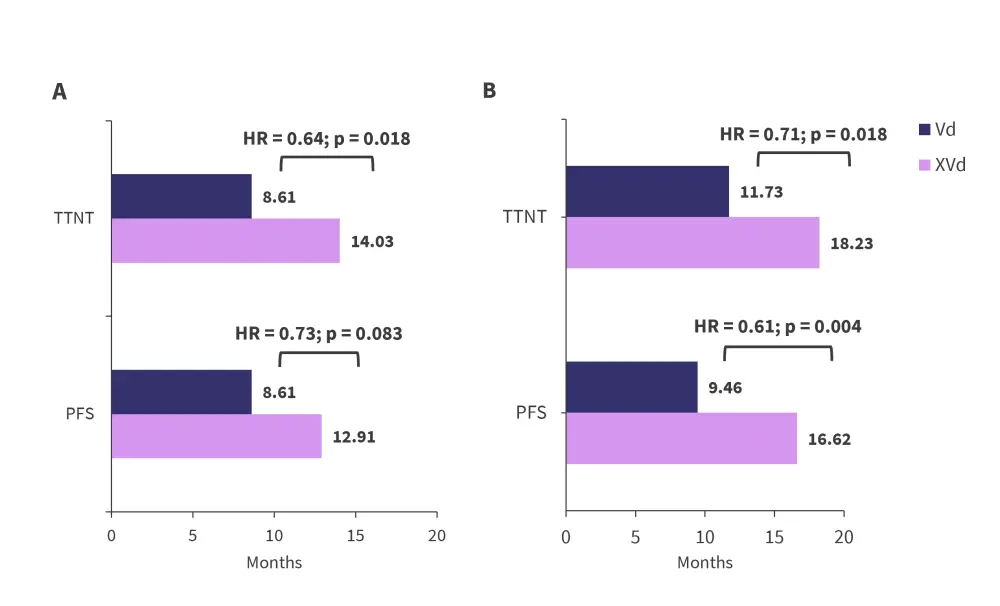
HR, hazard ratio; PFS, progression-free survival; TTNT, time to next treatment; Vd, bortezomib + dexamethasone; XVd, selinexor + bortezomib + dexamethasone.
*Adapted from Richard, et al.3
The overall response rate was higher in patients treated with XVd for both high- and standard-risk cytogenetics (Figure 2). Despite achieving very similar response rates and median duration of response, the differences were significant for longer-term outcomes and XVd did not overcome the poorer prognosis of high-risk cytogenetics.
Figure 2. Response rates in A high-risk patients and B standard-risk patients*
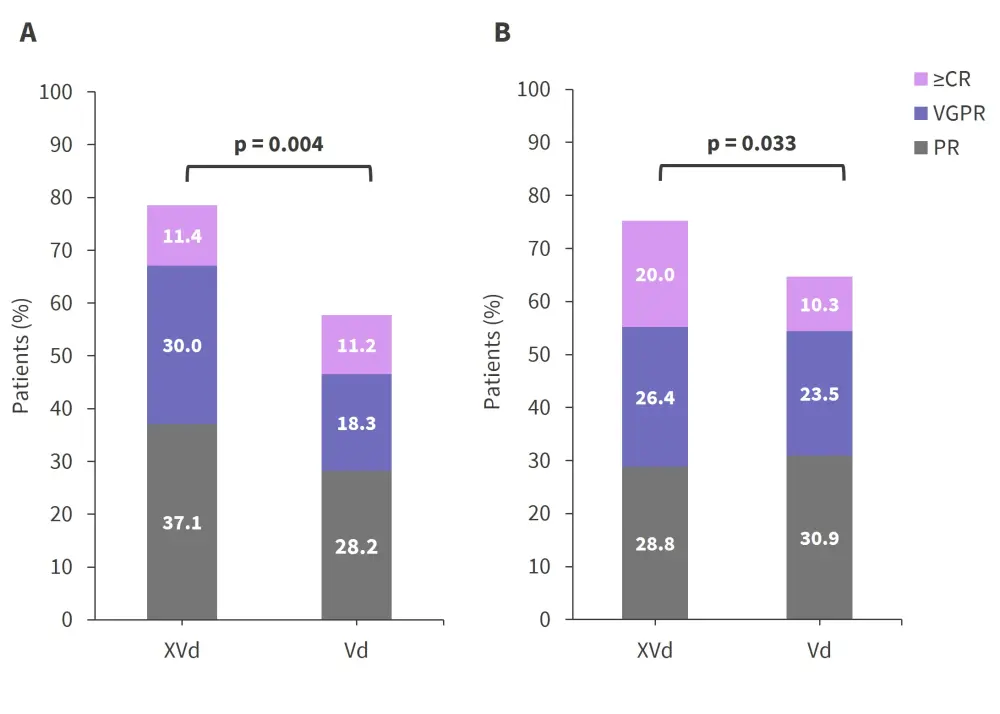
CR, complete response; MR, marginal response; PR, partial response; SD, stable disease; Vd, bortezomib + dexamethasone; VGPR, very good partial response; XVd, selinexor + bortezomib + dexamethasone.
*Adapted from Richard, et al.3
The median OS for XVd-treated patients with high-risk cytogenetics was 22.87 months compared with 24.84 months for Vd. The median OS was not reached for patients with standard-risk cytogenetics on either treatment combination. Moreover, XVd had lower mortality rates compared with Vd in patients with high-risk cytogenetics at 38.6% and 43.7%, respectively. In patients with standard-risk cytogenetics treated with XVd, the mortality rate was 21.6% compared with 28.7% for patients treated with Vd.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

