All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Safety and efficacy of the GPRC5D-targeted CAR T-cell therapy MCARH109
B-cell maturation antigen (BCMA)-targeted chimeric antigen receptor (CAR) T-cell therapy has shown deep and durable responses in patients with multiple myeloma (MM); however, patients are likely to relapse.1
G protein-coupled receptor family C group 5 member D (GPRC5D) is an orphan G protein-coupled receptor expressed in several myeloma cell lines and bone marrow (BM) plasma cells in patients with MM. Preclinical studies have demonstrated the efficacy of GPRC5D-targeted CAR T-cells, including activity in a BCMA antigen escape model.1
The Multiple Myeloma Hub has previously reported the efficacy of OriCAR-017, a second-generation, autologous, GPRC5D-directed CAR T-cell therapy. Here, we summarize the findings of a study by Mailankody, et al.2 that was published in the New England Journal of Medicine and reported the efficacy of MCARH109, a GPRC5D-targeted CAR T-cell therapy.
Study design
The study was an open-label, phase I, single-center, dose-escalation trial (NCT04555551) in heavily pretreated patients aged ≥18 years with relapsed/refractory MM, who had measurable disease, and an Eastern Cooperative Oncology Group performance status score of 0 or 1. Eligible patients underwent apheresis and lymphodepletion followed by a dose escalation phase with a single infusion of MCARH109 (Figure 1). Patients were followed until disease progression, death, or withdrawal.
Figure 1. Study design*
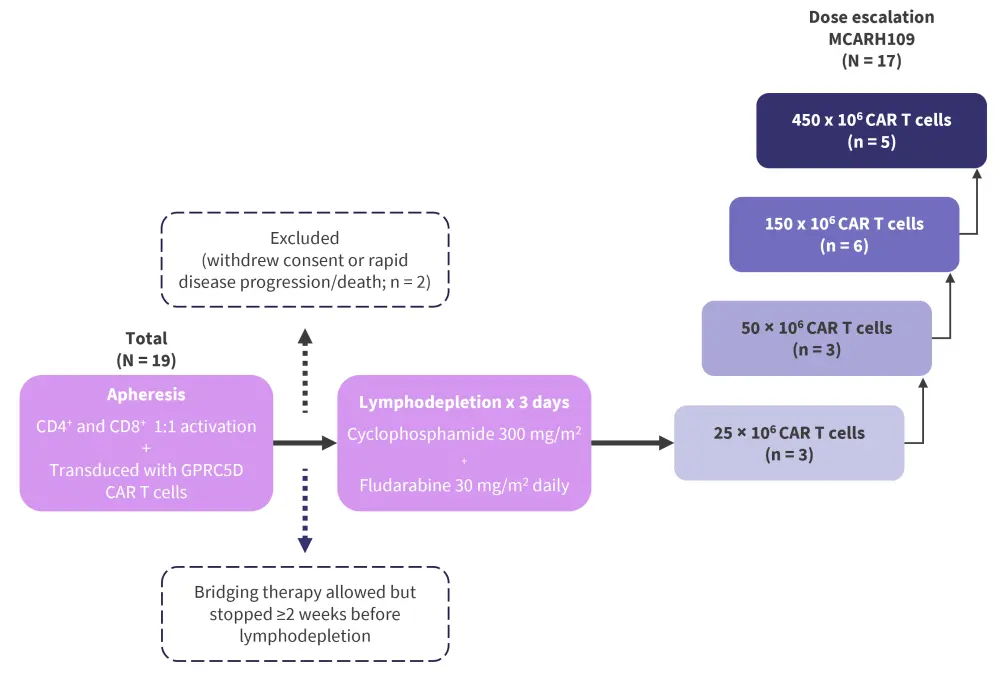
CAR, chimeric antigen receptor; GPRC5D, G protein-coupled receptor family C group 5 member D.
*Data from Mailankody, et al.2
The primary endpoint was the safety of MCARH109 and included assessing the following:
- Adverse events (AEs); graded based on the National Cancer Institute Common Terminology Criteria for AEs
- Cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS); graded according to the American Society for Transplantation and Cellular Therapy consensus guidelines
Key secondary endpoints included clinical response based on the response assessment criteria by the International Myeloma Working Group and minimal residual disease in BM assessed by flow cytometry. Exploratory endpoints included quantification of MCARH109, immunophenotyping, and GPRC5D immunohistochemical analysis.
Results
Baseline characteristics
A total of 17 patients were included, with a median age of 60 years (range, 38–76 years) and 76% of patients were male. Overall, 14 patients (82%) were refractory to their last line of therapy and 16 patients (94%) received bridging therapy after leukapheresis (Table 1).
Table 1. Baseline characteristics*
|
auto, autologous; allo, allogeneic; BCMA, B-cell maturation antigen; CAR, chimeric antigen receptor. |
|||||
|
Characteristics, % (unless stated otherwise) |
25 × 106 CAR T cells (n = 3) |
50 × 106 CAR T cells (n = 3) |
150 × 106 CAR T cells (n = 6) |
450 × 106 CAR T cells (n = 5) |
Total (N = 17) |
|---|---|---|---|---|---|
|
High-risk cytogenetic feature† |
100 |
67 |
50 |
100 |
76 |
|
Extramedullary plasmacytoma |
100 |
33 |
67 |
0 |
47 |
|
Non-secretory myeloma |
67 |
0 |
17 |
0 |
18 |
|
Refractory to the last line of therapy |
100 |
100 |
83 |
60 |
82 |
|
Triple refractory disease‡ |
100 |
100 |
100 |
80 |
94 |
|
Prior therapies, median (range) |
6 (6–8) |
5 (4–8) |
7 (5–14) |
6 (5–12) |
6 (4–14) |
|
Penta-exposed§ |
100 |
100 |
100 |
100 |
100 |
|
Auto transplantation |
0 |
67 |
17 |
0 |
18 |
|
Allo transplantation |
0 |
67 |
17 |
0 |
18 |
|
BCMA therapy¶ |
33 |
33 |
67 |
80 |
59 |
|
CAR T-cell therapy |
0 |
33 |
50 |
80 |
47 |
|
Bridging therapy |
100 |
100 |
100 |
80 |
94 |
|
Refractory to |
100 |
100 |
83 |
80 |
94 |
Safety of MCARH109
Although one or more AEs were reported in all patients, most were Grade 1 or 2. Hematologic AEs were the most common Grade 3 or 4 events, with selected AEs shown in Figure 2. CRS was observed in 15 patients of Grade 1 or 2, except one patient who received the highest dose (450 x 106 CAR T cells) and had Grade 4 CRS, ICANS, and macrophage activation syndrome. This patient received siltuximab and anakinra, in addition to tocilizumab and dexamethasone, for the management of CRS.
Figure 2. Safety of MCARH109*
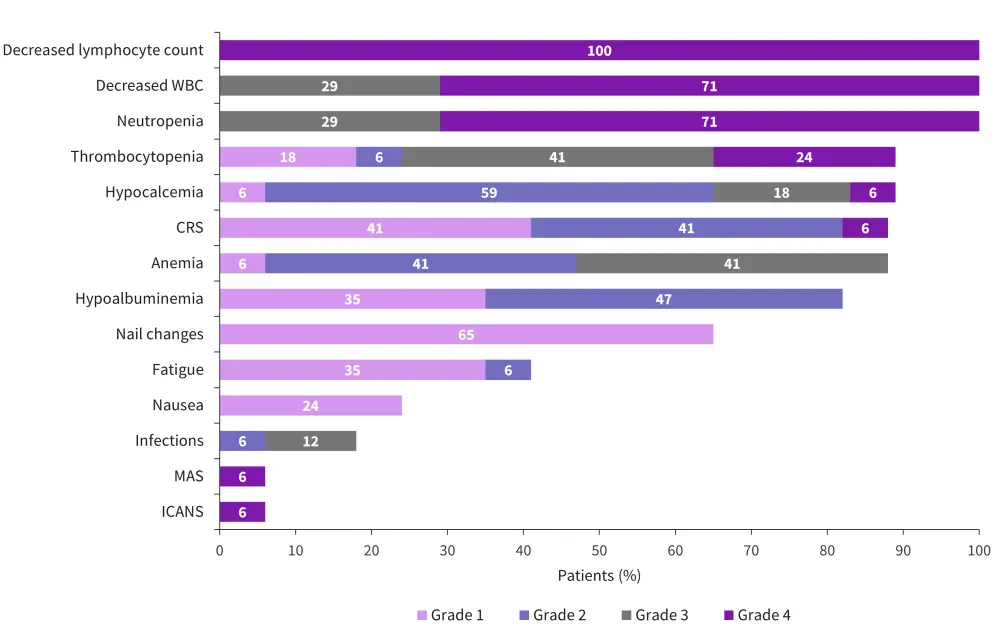
CRS, cytokine release syndrome; ICANS, immune effector cell-associated neurotoxicity syndrome; MAS, macrophage activation syndrome; WBC, white blood cells.
*Data from Mailankody, et al.2
Two additional patients who received the highest dose developed dizziness and unsteady gait, respectively, at 2.1 weeks after the MCARH109 infusion. These two patients also developed Grade 3 cerebellar disorder at 6.5 and 8.4 months, respectively. Magnetic resonance imaging (MRI) and repeat MRI plus positron-emission tomography within 6 weeks of symptom onset and 4 months, respectively, were negative for these two patients. Flow cytometric assessment of cerebrospinal fluid revealed a very low level of CAR T cells in one patient. At 7.7 months and 10.8 months of follow-up, respectively, both patients continue to have an ongoing Grade 3 cerebellar disorder. No other patient developed similar symptoms during a minimum of 6 months follow-up.
Clinical responses
A total of 71% (95% confidence interval (CI), 44–90) of patients showed a response across all dose levels of MCARH109, with a median duration of response of 7.8 months (95% CI, 5.7 to not reached) for all patients. The median duration of response was also 7.8 months (95% CI, 4.6 to not reached) in patients who received 25 × 106 to 150 × 106 CAR T cells. Figure 3 shows the clinical responses in all patients, including those who had received and not received prior BCMA therapies.
Figure 3. Clinical responses*
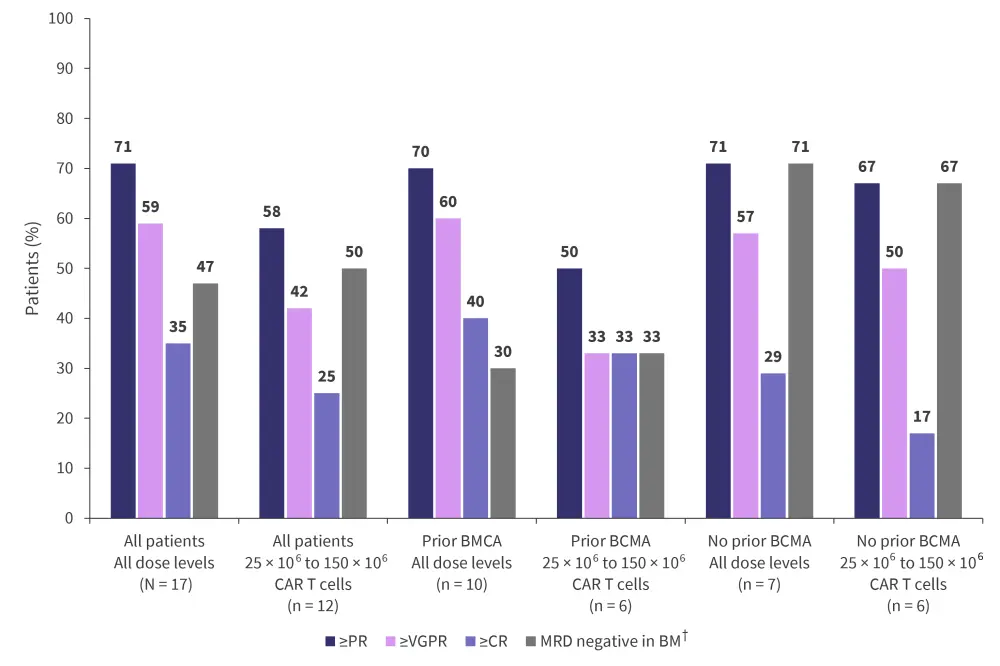
BCMA, B-cell maturation antigen; BM, bone marrow; CAR, chimeric antigen receptor; CR, complete response; MRD, minimal residual disease; PR, partial response; VGPR, very good partial response.
*Data from Mailankody, et al.2
†MRD assessed by flow cytometry (sensitivity, 10−5) at 4 weeks after infusion, at complete response, and as clinically indicated.
Quantification of MCARH109 showed robust expansion in the peripheral blood (PB) at all dose levels. The median peak expansion duration for all dose levels in PB was 14 days (range, 7–111 days) and the median area under the curve (AUC) during the first 28 days was 5,039,204 copies/mL. Both the peak expansion and AUC0–28 days were dose-dependent. MCARH109 in PB continued to be detected in 17, 11, and 1 patient at 4, 24, and 52 weeks, respectively.
Immunohistochemical analysis
GPRC5D was expressed in 88% of patients in their baseline BM or plasmacytoma biopsy. Four of the six patients who had progression after an initial response did not show GPRC5D expression at the time of progression and the remaining two had low levels of GPRC5D.
Conclusion
This study demonstrated that GPRC5D is an active target for MM even at a low dose of 25 × 106 CAR T cells. Dose-limiting toxic effects were seen in three patients, including two with persistent cerebellar disorders, who received the highest dose of 450 × 106 CAR T cells. However, no dose-limiting toxic effect or cerebellar disorders occurred at dose levels 25 × 106 or 150 × 106 CAR T cells; therefore, 150 × 106 CAR T cells was the maximum tolerated dose. The study also showed that clinical responses were positive, particularly in patients who were previously treated and refractory to BCMA CAR T-cell therapy. Overall, the findings support the concept that GPRC5D is a new and effective target for immunotherapy in patients with MM. Further studies to evaluate the safety of MCARH109 at dose level 450 × 106 CAR T cells are needed. NCT04674813 is an ongoing, multicenter trial of GPRC5D-targeted CAR T-cell therapy.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content


