All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Molecular-oriented therapy approaches in patients with RRMM
Multiple myeloma (MM) is a malignant disease characterized by clonal proliferation of abnormal plasma cells.1 Over the past two decades, the treatment landscape for MM has significantly advanced, including the development of next-generation sequencing techniques. As a result, actionable mutations such as BRAF can be identified and targeted for potentially tailored therapies.1 For a greater overview of the current landscape of precision medicine in MM, check out our previous article highlighting current targeted treatments and future possible targets.
Andreozzi et al.1 recently published results from the multiple myeloma early phase I (MM-EP1) study assessing the clinical benefits of personalized molecular-oriented (MO) versus non-molecular-oriented (no-MO) approaches in patients diagnosed with relapsed/refractory (R/R) MM. We summarize the key findings here.
Study design
This was a retrospective study analyzing data collected between 2013 and 2022 from 103 patients with R/R MM with at least one prior line of treatment. The study criteria for molecular characterization were as follows:
- Age ≥18 years
- Confirmed R/R MM
- Performance status 0–2
- Measurable disease in serum
- Consent for tumor molecular analysis and constitutional DNA analysis
Patients were considered as treated with a MO approach if they presented with t(11;14) and treated with BCL2 inhibitors, t(4;14) with FGFR3 rearrangement and treated with FGFR3 inhibitors, or a BRAF mutation and treated with BRAF inhibitors. Patients treated with therapies without an MO approach were included in the no-MO group. The efficacy endpoints included tumor response rate, overall survival, progression-free survival, and percentage reduction in monoclonal component.
Results
Baseline characteristics
The median age of patients was 67 years (range, 44–85 years) and the median number of previous lines of therapy was 4 (range, 1–8; Table 1).
Table 1. Patient characteristics*
|
ISS, International Staging System; MO, molecular oriented; NA, not available; no-MO, non-molecular oriented. |
||
|
Characteristic, % (unless stated otherwise) |
MO |
No-MO |
|---|---|---|
|
Sex, n |
|
|
|
Male |
7 |
49 |
|
Female |
10 |
37 |
|
Median age (range), years |
59 (48–81) |
68 (44–85) |
|
ISS |
||
|
I |
29 |
28 |
|
II |
41 |
17 |
|
III |
12 |
26 |
|
NA |
18 |
29 |
|
Cytogenetic risk |
|
|
|
t(4;14) |
24 |
20 |
|
t(11;14) |
53 |
12 |
|
Chromosome 1 |
24 |
20 |
|
del13q14 |
6 |
2 |
|
del17p |
18 |
11 |
|
NA |
12 |
11 |
|
Median number of prior lines (range), n |
5 (1–11) |
3 (1–8) |
Molecular characteristics
A total of 93 patients were included in the cytogenetic analysis. Figure 1 shows the most recurrent cytogenetics alterations.
Figure 1. Most recurrent cytogenetic alterations*
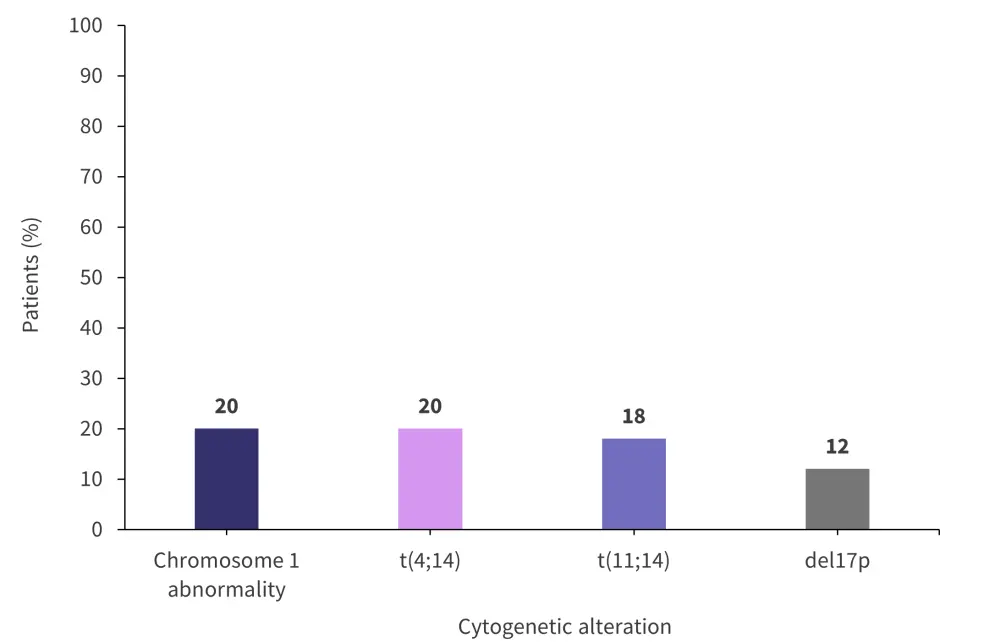
*Adapted from Andreozzi, et al.1
The median number of aberrant genes identified by next-generation sequencing analysis was 2 (range, 1–7) and the distribution of these gene mutations is shown in Figure 2.
Figure 2. Gene mutations identified by NGS*
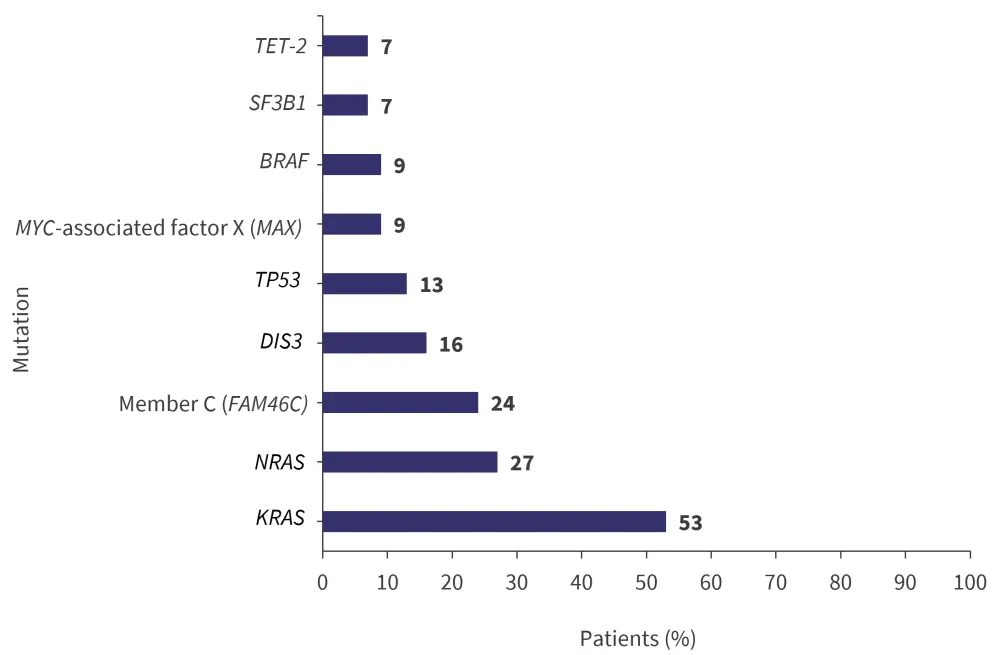
NGS, next-generation sequencing.
*Adapted from Andreozzi, et al.1
The mutational interaction matrix identified a significant co-existence of NRAS and FAM46C mutations (p < 0.01) and a significant mutual exclusion of KRAS and NRAS mutations (p < 0.05). DIS3 and KRAS mutations were mostly associated with a chromosome 1 abnormality, while BRAF and NRAS mutations were associated with a normal karyotype.
Potentially actionable molecular targets were identified in 48% of patients (Figure 3). The median variation in measurable monoclonal component in serum was a reduction of 91% vs 30% (p = 0.33) in the MO and no-MO groups.
Figure 3. Potential actionable molecular targets*
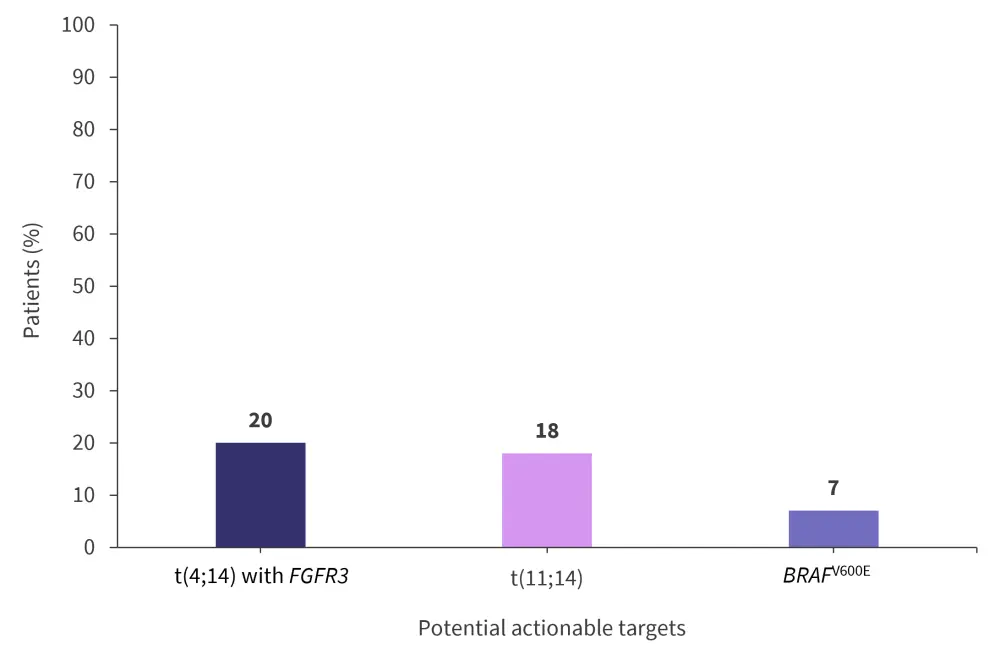
*Adapted from Andreozzi, et al.1
Efficacy endpoints
The overall response rate was 65% vs 58% in the MO and no-MO groups, respectively (Figure 4). The median progression-free survival was 9 months vs 6 months (hazard ratio, 0.96; 95% confidence interval, 0.51–1.78; p = 0.88) and the median overall survival was 26 months vs 28 months (hazard ratio, 0.98; 95% confidence interval, 0.46–2.12; p = 0.98) in the MO and no-MO groups, respectively.
Figure 4. Response rates*
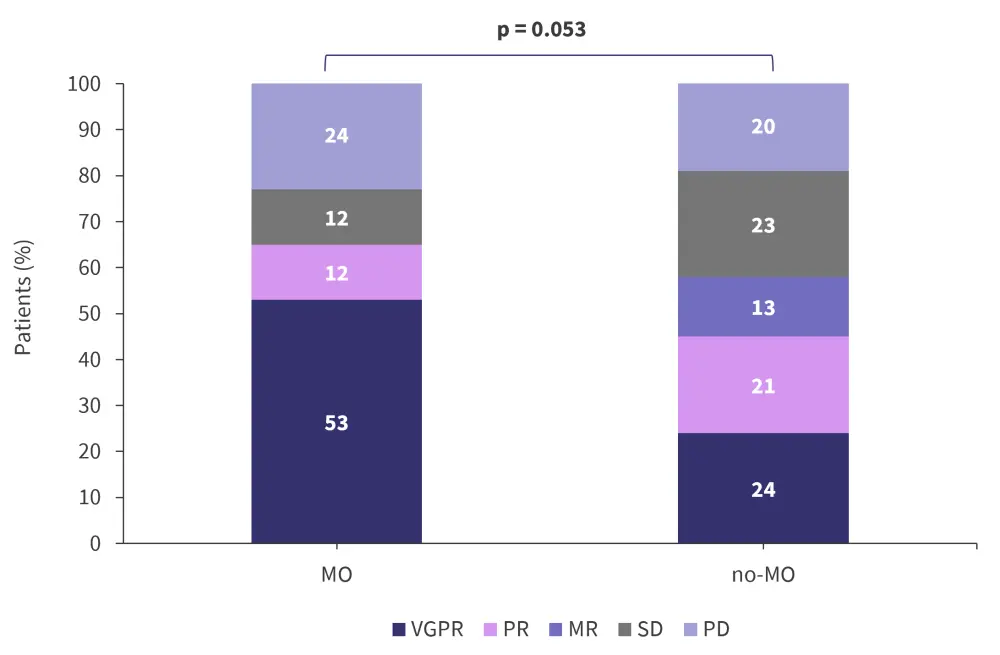
MO; molecular oriented; MR, minimal response; no-MO, non-molecular oriented; PD, progressive disease; PR, partial response; SD, stable disease; VGPR, very good partial response.
*Adapted from Andreozzi, et al.1
Overall, 54 deaths occurred, eight in the MO group and 46 in the no-MO group. The causes of death included disease progression (n = 47), secondary malignancy (n = 3), COVID-19 (n = 2), influenza infection (n = 1), and coronary ischemic disease (n = 1).
Conclusion
This retrospective study highlights the potential relationships between genetic mutations and karyotype abnormalities. However, the benefits of MO treatment remain unclear as it did not translate into improved clinical outcomes compared with no-MO therapies. The study was limited by its retrospective nature, small sample size, patient selection bias, and the sequencing panel limit of 29 genes, thus, not allowing a complete characterization of the R/R MM samples. Therefore, future studies on the use of genomics and improving algorithms of molecular precision medicine for patients with MM are warranted.
Your opinion matters
Would you consider using molecular oriented-approaches compared with non-molecular oriented treatments?
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content



