All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the International Myeloma Foundation or HealthTree for Multiple Myeloma.
The Multiple Myeloma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Multiple Myeloma Hub cannot guarantee the accuracy of translated content. The Multiple Myeloma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Multiple Myeloma Hub is an independent medical education platform, sponsored by Bristol Myers Squibb, GSK, Legend Biotech, Pfizer, and Roche. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View multiple myeloma content recommended for you
Extended follow-up from the DREAMM-2 trial
The potential of belantamab mafodotin (belamaf) in the relapsed or refractory (R/R) multiple myeloma (MM) setting was highlighted in 2019, when the pivotal DREAMM-2 study met its primary endpoint. The anti-B-cell maturation antigen (BCMA) antibody-drug conjugate demonstrated clinically meaningful overall response rates (ORRs), and in 2020 received approval from the U.S. Food and Drug Administration (FDA) and conditional marketing authorization from the European Commission.
Sagar Lonial and colleagues1 recently published the results from a long-term analysis of the pivotal DREAMM-2 study (NCT03525678), evaluating single-agent belamaf for the treatment of patients with R/R MM who
- have received ≥3 lines of therapy;
- are refractory to an immunomodulatory agent and a proteasome inhibitor; and
- are refractory and/or intolerant to an anti-CD38 monoclonal antibody.
The Multiple Myeloma Hub is happy to present a summary of the 13-month follow-up of the DREAMM-2 study. Click here for a comprehensive overview of the clinical history of belamaf, the DREAMM-2 study design, and primary analysis efficacy and safety data. Why not also take a look at a summary of the DREAMM trials from the 2020 American Society of Clinical Oncology (ASCO) Annual Meeting, where data from the DREAMM-2 trial were considered practice changing.
Results
- Latest data cutoff: January 31, 2020.
- Patients remaining on the approved dose of belamaf, 2.5 mg/kg: 10%
- Responses and survival outcomes of patients enrolled to receive 2.5 mg/kg belamaf are outlined in Table 1.
- Among the 31 patients who achieved a partial response or better
- 18 patients (58%) achieved a very good partial response or better
- 7 patients achieved a stringent complete response or complete response
- Median overall survival (OS): not reached (NR)
- Median duration of response: 13.1 months (4.9 months–NR)
- 1-year OS: 87% (69–95%)
Table 1. Patient responses to 2.5 mg/kg belamaf in the DREAMM-2 trial*
|
Belamaf, belantamab mafodotin; CBR, clinical benefit rate; CI, confidence interval; CR, complete response; DoR, duration of response; ITT, intention to treat; MR, minimal response; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; sCR, stringent complete response; SD, stable disease; VGPR, very good partial response. |
|
|
Independent Review Committee-Assessed Best Response, %† |
ITT population |
|
ORR, % |
32 |
|
≥CR, % |
7 |
|
VGPR, % |
11 |
|
PR, % |
13 |
|
MR, % |
4 |
|
SD, % |
28 |
|
CBR, % |
36 |
|
Median DoR, months |
11.0 |
|
Median OS, months |
13.7 |
|
Median PFS, months |
2.8 |
- ORRs were comparable across patients with varying renal function and cytogenetic risk but were inferior in patients with extramedullary disease (Figure 1).
- No additional safety concerns were observed, and incidence was comparable between subgroups.
- Treatment‐emergent adverse events (TEAEs): 88%
- TEAEs Grade 3–4: 57%
- Most common Grade ≥3 adverse events (AEs): keratopathy, thrombocytopenia, and anemia.
- At data cutoff, 77% of patients with keratopathy had recovered.
- Dose delays and reductions due to AEs (mainly due to keratopathy) were reported in 54% and 35% of patients, respectively. Of note, they had minimal impact on patient response.
- 9% discontinued permanently due to AEs.
- Treatment‐emergent adverse events (TEAEs): 88%
Figure 1. ORRs in the overall and subgroup populations of patients in the 2.5 mg/kg belantamab mafodotin cohort of the DREAMM-2 trial*
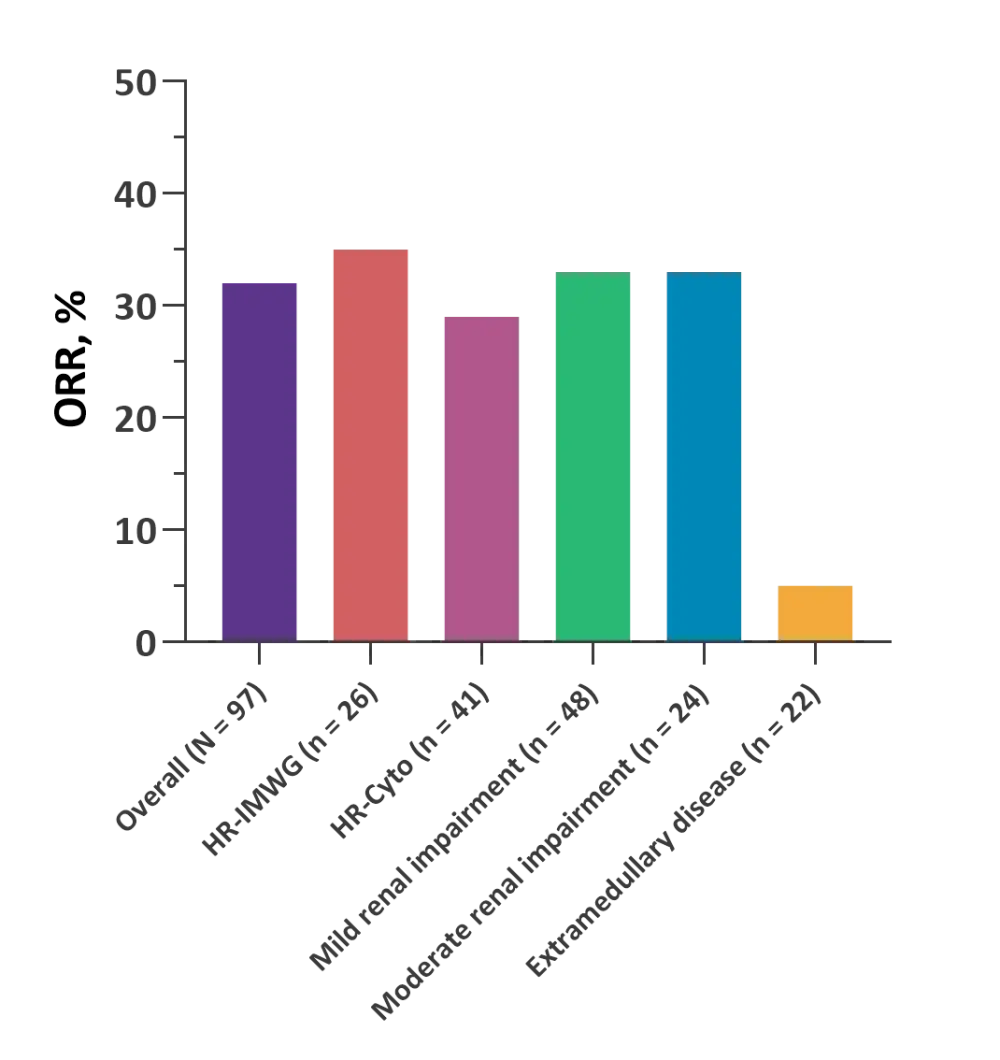
HR-cyto, high-risk cytogenetics, including 1q21 gain/amplification; HR-IMWG, high-risk cytogenetics according to International Myeloma Working Group criteria; ORR, overall response rate.
*Data from Lonial S, et al.1
Conclusion
Data from the longer-term analysis highlight the durable clinical efficacy of belamaf in patients with heavily pretreated R/R MM. No additional safety concerns were associated with belamaf in this patient population.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

